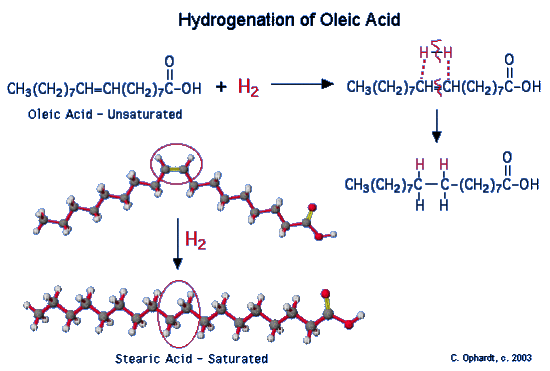Unsaturated fatty acids can enter in different chemical reactions that alter their structure and characteristics leading to denaturation and changes of the content of the oils or foods they are part of. Food processing industry uses those different reactions in order to change their aggregate state and get the required refined products of fats and oils that have a specific density, color, smell or taste that differs from the natural. But, what do those processes of refining of the oils truly mean? Are they really just changing the oil’s color or taste, or are an essential denaturation of the oils which converts them into synthetic forms that have negative impact on the human metabolism and health?
The process of refining of the oils, in its broader content starts with the process of oil extraction. Plant oils for the processing industry are mainly obtained from sunflower, soy, peanuts, canola and other seeds and plants. First the seeds are grinded under pressure and the oil is being extracted. To achieve complete extraction after pressing, seeds are being steam boiled with some chemical solvents such as hexane trichlorethylene and acetone (both toxic and highly inflammable petroleum derivatives). After the extraction the refining continues by elimination of the unwanted elements (color, aroma) or compounds that may spoil the oil. This procedure is performed by adding sodium hydroxide and heating up the oil at a temperature of 200˚C. In addition, in order to have complete control of the oil’s content, it is processed with carbon that removes vitamins, natural lecithin, chlorophyll and other micronutrients that have significant nutritional value in the natural oil!
The process of hydrogenation of the fatty acids and triglycerides is a reaction of saturation of the unsaturated carbon atoms. Basically, as an industrial process it is used as a method for “hardening” of the fatty acids. The hydrogenation enables hydrogen ions to be attached to the unsaturated positions along the carbon chain of the fatty acid molecule, which results in flattening of the molecular structure, thus transforms the oil to a semisolid or solid state product at room temperature. This procedure is most often used in the production of margarine and other solid products made out of plant oils. Specifically, after the oil is refined, using a catalyst (nickel or aluminum ions 0,01-0,02%), the oil is put under high pressure and heat (2-10 atmospheres, 160-220°C) and bombarded with hydrogen ions. The hydrogen ions enter in reactions with the unsaturated carbon atoms creating artificial trans bonds within the fatty acid, which is the reason why these types of fats are named trans fats. The content of hydrogenated, trans fats in processed foods can be up to 40%, which makes those products and their consumption bad for the health.
In addition to hydrogenation, hardening of the liquid plant oil could be achieved with the process of inter esterification. In this process an enzyme called lipase, isolated from a bacteria or fungi), is being added to the oil to separate the fatty acids present in the oil’s triglycerides from the triglycerole (alcohol) backbone of the molecule. The end goal of the process is achieving recombination the unsaturated and saturated fatty acids, i.e. reorder of the fatty acids in the triglyceride molecule that will change its melting point and state at room temperature.
During the last phase of oil processing the manufacturers “improve” the organoleptic properties of the oil by changing the oil’s appearance, texture and taste. The denaturation of the natural structure of the oils and the formation of trans fats generates bad odor that is removed by steaming. Additional chemicals are also added to bleach out the product. Finally, in order to satisfy the consumer demands and formal dietary recommendations, the manufacturers finish the process of refining of the oils by fortifying the final product with vitamins or omega 3 fatty acids, nutrients that are already present and abundant in the natural oil, yet have been destroyed during its processing!
Unlike these processed, refined oils, cold pressed oils are obtained in a process of cold mechanical extractions at temperatures below 45°C. This allows preservation of all fatty acids, vitamin E, omega 3 fatty acids, sterols and other important nutrients found in oils in their natural, bioactive form that benefits our metabolism and supports our health.